How to Verify THC Products Are Federally Compliant Before Buying
Posted by Enjoy Hemp Editor Team on Feb 19th 2026
Last Updated: February 2026
Purchasing hemp-derived THC products without understanding federal compliance requirements puts you at risk of buying illegal substances, wasting money on mislabeled products, or encountering legal issues. The regulatory landscape has shifted dramatically, making verification more important than ever.
In my 15 years evaluating wellness products, I have witnessed the hemp market transform from a niche industry to a complex regulatory environment where consumers must become their own compliance officers. The tools and knowledge needed to verify federal compliance are straightforward once you understand what to look for.
| Compliance Factor | Current Requirement (2018 Farm Bill) | Upcoming Requirement (Nov 2026) |
|---|---|---|
| THC Threshold | ≤0.3% delta-9 THC by dry weight | ≤0.3% total THC (including THCA) by dry weight |
| Final Product Limit | Must meet dry weight calculation | ≤0.4mg total THC per container |
| Synthetic Cannabinoids | Legal gray area for converted cannabinoids | Excluded from hemp definition |
| Lab Testing | Required for cultivation; recommended for products | FDA guidance expected on testing standards |
| Hemp Source | Must be from Cannabis sativa L. | Naturally produced cannabinoids only |
| Documentation | COA with batch-specific results | Enhanced traceability requirements likely |
What Makes a THC Product Federally Compliant Under Current Law?
A THC product is federally compliant under the 2018 Farm Bill when it is derived from hemp (Cannabis sativa L.) and contains no more than 0.3% delta-9 THC on a dry weight basis. This calculation applies to the finished product, not just the source plant, meaning manufacturers must formulate products to meet this threshold after processing.
When I led product development at a national supplement brand, understanding the dry weight calculation became essential for creating compliant formulations. The math determines whether a product falls under legal hemp or controlled marijuana classification.
According to the Congressional Research Service analysis of the 2018 Farm Bill, hemp is defined as "the plant Cannabis sativa L. and any part of that plant, including the seeds thereof and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol concentration of not more than 0.3 percent on a dry weight basis."
This definition created what many call the "Farm Bill loophole" because it specifically references delta-9 THC rather than total THC. Products containing other cannabinoids like delta-8 THC, delta-10 THC, or THCA could technically qualify as hemp if delta-9 levels remained below 0.3%.
For consumers, federal compliance verification starts with checking the Certificate of Analysis (COA) for delta-9 THC percentage. Reputable brands like Enjoy Hemp publish batch-specific COAs showing exact cannabinoid concentrations, making compliance verification straightforward.
How Do You Calculate the 0.3% THC Dry Weight Limit?
The 0.3% dry weight calculation divides the amount of delta-9 THC by the total dry weight of the product, then multiplies by 100. For a compliant product, this percentage must equal 0.3% or less. This means a 3,500mg (3.5 gram) gummy can legally contain up to 10.5mg of delta-9 THC while remaining federally compliant.
After testing dozens of hemp products throughout my career, I have seen many consumers misunderstand this calculation. The dry weight measurement removes all moisture content, providing a consistent baseline for comparison across different product types.
Here is how the math works in practice:
Step 1: Identify the total dry weight of the product in milligrams or grams.
Step 2: Determine the delta-9 THC content from the COA.
Step 3: Divide THC content by total dry weight, then multiply by 100.
Example: A gummy weighing 4 grams (4,000mg) dry weight containing 10mg of delta-9 THC: (10 ÷ 4,000) × 100 = 0.25%. This product is compliant.
According to the USDA hemp program regulations, labs must use post-decarboxylation testing methods that account for the conversion of THCA to THC. The formula used is: Total THC = (0.877 × THCA) + THC.
This calculation matters because heavier products can contain more total milligrams of THC while staying within the percentage threshold. A 5mg THC beverage in a large bottle meets compliance differently than a small concentrate product.
What Documents Should You Check Before Purchasing THC Products?
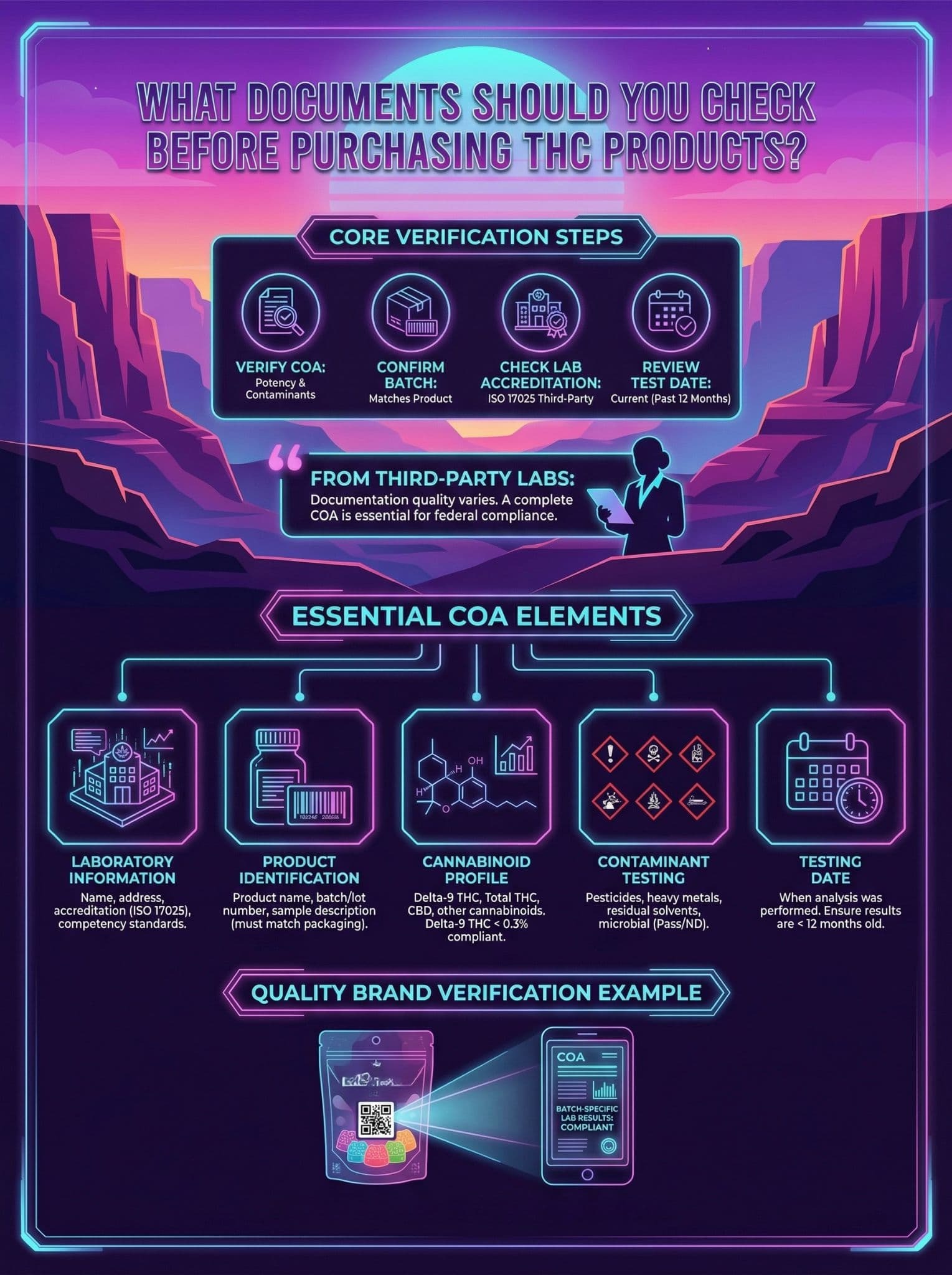
Before purchasing any THC product, verify the Certificate of Analysis (COA) showing cannabinoid potency and contaminant testing, confirm the batch number matches your specific product, check that an ISO 17025 accredited third-party laboratory performed the testing, and review the test date to ensure results are current within the past 12 months.
From my conversations with third-party testing labs, I have learned that documentation quality varies dramatically across the industry. A complete COA tells you everything needed to verify federal compliance.
Essential COA Elements:
- Laboratory Information: Name, address, and accreditation credentials of the testing facility. Look for ISO 17025 certification, which ensures the lab meets international competency standards.
- Product Identification: Product name, batch or lot number, and sample description. This must match exactly what appears on your product packaging.
- Cannabinoid Profile: Detailed breakdown of delta-9 THC, total THC, CBD, and other cannabinoids. The delta-9 THC percentage should clearly show compliance with the 0.3% threshold.
- Contaminant Testing: Results for pesticides, heavy metals, residual solvents, and microbial screening. All categories should show "Pass" or "ND" (not detected).
- Testing Date: When the analysis was performed. Results older than 12 months may not reflect current product quality.
Quality brands make COA verification simple. Companies like Enjoy Hemp's gummy collection include QR codes on packaging that link directly to batch-specific lab results, eliminating guesswork about which COA applies to your product.
How Can You Verify a COA Is Authentic and Not Fabricated?
Verify COA authenticity by confirming the testing laboratory exists as an independent, accredited facility, calling the lab directly using contact information from their official website (not the COA), checking that the batch number on the COA matches your product packaging exactly, and ensuring the QR code links to the laboratory's domain rather than the brand's website.
Based on my experience working with third-party testing labs, I recommend treating COA verification as a multi-step process. Fraudulent documentation exists in the market, making independent verification essential.
Verification Steps:
Step 1: Confirm Laboratory Existence
Search the laboratory name online. Legitimate labs have their own websites, physical addresses, and verifiable accreditation status. Check accreditation databases maintained by organizations like A2LA (American Association for Laboratory Accreditation).
Step 2: Cross-Reference Contact Information
The phone number and address on the COA should match information on the laboratory's official website. If they differ, the COA may be altered or fabricated.
Step 3: Match Batch Numbers
The batch or lot number on your product packaging must appear identically on the COA. Any discrepancy means that specific COA does not apply to your product.
Step 4: Check the QR Code Destination
Scan the QR code and note where it directs you. Legitimate COAs link to the testing laboratory's domain (like "labname.com/results") rather than the brand's own website. If the QR code leads to the brand's site hosting a PDF, additional verification is warranted.
Step 5: Look for Red Flags
Watch for missing contaminant testing, perfectly round percentages (like "exactly 20.00%"), no laboratory contact information, or results that seem too good given product pricing.
Which States Have Stricter Rules Than Federal Law?
Several states have enacted restrictions stricter than federal hemp law, including complete bans on intoxicating hemp products (Idaho, some aspects of California), bans on specific cannabinoids like delta-8 THC (Alaska, Arizona, Arkansas, Colorado, and others), and potency limits or sales restrictions (Alabama, Louisiana, many others). Always verify state laws for your specific location before purchasing.
The federal 0.3% delta-9 THC threshold establishes a floor, not a ceiling. States maintain authority to impose additional restrictions on hemp-derived products within their borders.
States with Complete or Near-Complete Bans:
- Idaho: Bans all THC-related cannabinoids, including those derived from hemp
- California: Prohibits intoxicating hemp products outside the regulated cannabis market
- Arkansas: Bans synthesized or converted cannabinoids including delta-8
States with Significant Restrictions:
- Alabama: Limits edibles to 10mg THC per serving, 40mg per package; bans smokable hemp
- Louisiana: Total THC calculation with 5mg per serving limits; bans smokable products
- Arizona: Requires sales through licensed dispensaries only
- Ohio: Ongoing regulatory disputes over intoxicating hemp products
Even federally compliant products may be illegal in your state. Before purchasing, verify your state's specific regulations regarding hemp-derived cannabinoids. Reputable brands typically block shipping to states where their products are not permitted.
What Changes Are Coming to Federal Hemp Compliance in 2026?

The FY2026 Agriculture Appropriations Act (H.R. 5371), signed November 12, 2025, redefines hemp using total THC (including THCA) rather than delta-9 THC alone, limits final products to 0.4mg total THC per container, and excludes synthetic or manufactured cannabinoids from the hemp definition. These changes take effect November 12, 2026.
In my 15 years evaluating wellness products, this represents the most significant regulatory shift since the 2018 Farm Bill legalized hemp. The changes will dramatically reshape what products qualify as legal hemp.
Key Changes Taking Effect November 2026:
Total THC Calculation: Hemp must now contain ≤0.3% total THC (including THCA) on a dry weight basis, not just delta-9 THC. This closes the loophole that allowed high-THCA products to qualify as hemp.
Per-Container Limits: Final hemp-derived cannabinoid products cannot exceed 0.4mg of combined total THC per container. This effectively eliminates most intoxicating hemp products currently on the market.
Synthetic Cannabinoid Exclusion: Cannabinoids that are "synthesized or manufactured outside the plant" are excluded from the hemp definition, even if derived from legal hemp. This targets delta-8 THC and similar converted cannabinoids.
FDA Guidance Required: The FDA must publish lists of naturally occurring cannabinoids, THC-class cannabinoids, and cannabinoids with similar effects to THC. These lists will determine which compounds count toward compliance thresholds.
Until November 2026, products meeting current 2018 Farm Bill standards remain federally legal. However, consumers should monitor legislative developments, as proposals to modify or repeal these changes have already been introduced.
How Do You Know If a Brand Prioritizes Federal Compliance?
Compliance-focused brands publish batch-specific COAs from ISO 17025 accredited laboratories, clearly display THC content per serving and per package, source from licensed U.S. hemp farms with documented chain of custody, respond transparently to compliance questions, and proactively communicate when regulations change.
When I led product development at wellness brands, we viewed compliance not as a burden but as a competitive advantage. Brands that invest in proper documentation demonstrate commitment to consumer safety and product quality.
Indicators of a Compliance-Focused Brand:
- Accessible Lab Results: COAs are easy to find, either on product pages or through scannable QR codes on packaging
- Complete Testing Panels: Testing includes cannabinoid potency plus full contaminant screening (pesticides, heavy metals, solvents, microbials)
- Batch Matching: Each product clearly displays a batch number that corresponds to specific COA documentation
- Transparent Sourcing: Information about hemp origin, cultivation practices, and extraction methods is readily available
- Regulatory Awareness: The company acknowledges shipping restrictions and state-specific limitations
Companies like Enjoy Hemp exemplify this approach, maintaining USDA-certified organic sourcing, comprehensive third-party testing, and clear communication about product compliance. Their THC-infused beverages demonstrate how brands can deliver enjoyable products while maintaining rigorous compliance standards.
What Should You Do If You Suspect a Product Is Non-Compliant?
If you suspect non-compliance, stop using the product immediately, contact the manufacturer to request current COA documentation, report concerns to your state's hemp program or attorney general office, and consider reporting to the FDA if the product makes unauthorized health claims or lacks required testing documentation.
From my work consulting for wellness publications, I have seen that non-compliant products pose real risks beyond legal concerns. Products exceeding THC thresholds may cause unexpected intoxication, while those with incomplete testing could contain harmful contaminants.
Signs of Potential Non-Compliance:
- No COA available or COA does not match product batch number
- Testing performed by in-house lab rather than independent third party
- Missing contaminant testing results
- THC content that seems inconsistent with reported effects
- Packaging that mimics name-brand candy or food products
- Prices significantly below market average for comparable products
Reporting Options:
State hemp programs maintain oversight of licensed producers and can investigate compliance concerns. The FDA handles complaints about products making unauthorized health claims or those that may pose safety risks. Many states have specific consumer protection hotlines for cannabis and hemp product concerns.
Frequently Asked Questions
Does Federal Compliance Guarantee a Product Is Legal in My State?
No. Federal compliance establishes the baseline, but states can impose stricter restrictions. A product meeting federal standards may still be illegal in states that ban certain cannabinoids, require sales through licensed dispensaries, or impose lower potency limits. Always verify your state's specific hemp product regulations before purchasing.
The patchwork of state regulations creates significant complexity for consumers. What you can legally purchase in Texas may be prohibited in California, despite both products meeting federal standards.
Can I Travel Across State Lines with Federally Compliant THC Products?
Traveling with hemp-derived THC products carries legal risk even when products meet federal standards. The destination state's laws apply upon arrival, meaning a federally compliant product could become illegal once you cross certain state borders. For air travel, TSA defers to local law enforcement on cannabis products. Avoid traveling with THC products unless you have verified legality in all jurisdictions.
Even with a valid COA, law enforcement may not distinguish between legal hemp and illegal marijuana during a traffic stop or airport screening.
How Often Should Brands Update Their COA Documentation?
Reputable brands test every production batch and maintain current COAs for all products in active inventory. COAs older than 12 months may not reflect current product quality due to potential degradation. Look for testing dates within the past year, and prefer brands that clearly date their COAs and test consistently.
Batch testing catches variations in cannabinoid content and ensures each product meets the same quality standards, providing consistency that periodic testing cannot guarantee.
What Is the Difference Between Delta-9 THC and Total THC?
Delta-9 THC refers specifically to the primary psychoactive cannabinoid in cannabis. Total THC accounts for delta-9 THC plus THCA (the acidic precursor that converts to THC when heated), using the formula: Total THC = (0.877 × THCA) + THC. Current federal law measures only delta-9 THC, but 2026 regulations will require total THC calculations.
This distinction matters significantly for compliance. A product could contain substantial THCA (which becomes psychoactive when smoked or vaped) while meeting current delta-9 limits, but would fail under total THC requirements.
Making Informed Compliance Decisions
Verifying federal compliance before purchasing THC products protects both your legal standing and your health. The process requires checking documentation, understanding calculations, and staying informed about evolving regulations.
Start with the COA: locate it, verify the laboratory's independence, confirm batch numbers match, and ensure delta-9 THC falls within the 0.3% threshold. Check your state's specific restrictions. And monitor the regulatory landscape as significant changes approach in November 2026.
Brands like Enjoy Hemp demonstrate that federal compliance and product quality work together. With USDA-certified organic sourcing, comprehensive third-party testing, and transparent documentation, consumers can enjoy hemp-derived products with confidence in their legal status and safety.
When in doubt, contact the brand directly with compliance questions. How they respond tells you much about their commitment to operating within legal boundaries and prioritizing consumer wellbeing.
By Sarah Mitchell, Wellness Industry Specialist
Sarah Mitchell is a wellness industry veteran with 15 years of experience in functional supplements and hemp-derived products. She previously led product development at two nationally recognized wellness brands and holds certification as a holistic health practitioner. Sarah now advises consumers on quality standards, helping them make informed decisions in the evolving hemp market.
Disclaimer: This article is for educational purposes only and does not constitute legal advice. Hemp and THC regulations vary by jurisdiction and change frequently. Consult with a legal professional for advice specific to your situation. Verify current federal and state laws before purchasing any THC products.

























